Complete Antiparasitic Cancer Protocol: Fenbendazole, Ivermectin & Mebendazole Explained

Drug repurposing has gained significant attention in oncology, with medications like fenbendazole, ivermectin, and mebendazole being widely discussed. Known as the antiparasitic cancer protocol, this approach is based on early scientific findings but remains unapproved for cancer treatment.
Understanding the difference between scientific evidence and anecdotal claims is critical when evaluating such therapies, especially in life-impacting conditions like cancer.
What Is the Antiparasitic Cancer Protocol?
The antiparasitic cancer protocol refers to the off-label use of three drugs:
- Fenbendazole (primarily used in veterinary medicine)
- Ivermectin (used in both humans and animals)
- Mebendazole (approved for parasitic infections in humans)
These drugs were originally developed to treat parasitic infections but are now being explored for potential anticancer effects such as:
- Inhibiting tumor growth
- Disrupting cancer metabolism
- Enhancing immune response
However, it is important to note that no major regulatory authority has approved this protocol for cancer treatment.
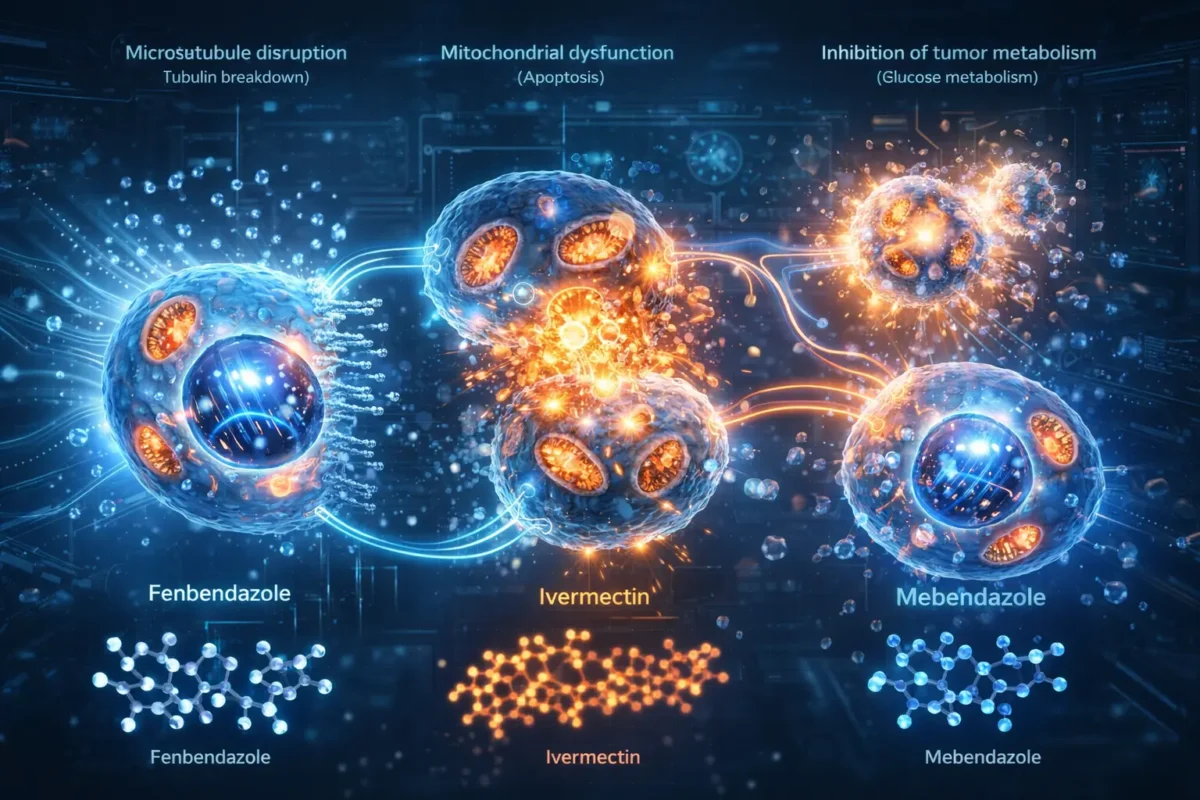
Mechanism of Action: How These Drugs May Work
Microtubule Disruption
Fenbendazole and mebendazole belong to the benzimidazole class and may interfere with microtubules, which are essential for cell division.
This may lead to:
- Inhibition of tubulin polymerization
- Arrest of the cell cycle
- Induction of apoptosis (programmed cell death)
Metabolic Interference (Warburg Effect)
Cancer cells rely heavily on glucose metabolism. Research suggests these drugs may:
- Reduce glycolysis
- Block glucose uptake
- Limit tumor energy supply
This could theoretically “starve” cancer cells.
Multi-Targeted Approach
The combination protocol is believed to act through multiple pathways, including:
- Structural disruption
- Metabolic interference
- Immune modulation
- Signaling pathway suppression
This multi-targeted concept makes the protocol appealing, though not clinically validated.
What Does Scientific Research Say?
Preclinical Evidence
Laboratory and animal studies have shown:
- Reduced cancer cell growth
- Increased apoptosis
- Slower tumor progression
- Reduced metastasis
However, these findings are preliminary and not sufficient for clinical application.
Clinical Trials Status
- Mebendazole: Early-stage trials (e.g., glioblastoma)
- Ivermectin: Limited preliminary research
- Combination protocol: No reliable human clinical trials
Overall, there is no strong clinical evidence supporting this protocol as a cancer treatment.
Risks and Safety Concerns
Unregulated Usage
- No standardized dosage
- No defined treatment duration
- Lack of safety guidelines
Potential Side Effects
Improper use may result in:
- Liver toxicity
- Drug interactions
- Delay in receiving proven treatments
There have been reported cases linking unsupervised use of fenbendazole to serious liver complications.
Veterinary vs Human Formulations
Fenbendazole is often sold for animal use, which raises concerns about:
- Purity
- Dosage accuracy
- Contamination
Why Is This Protocol Popular Online?
The growing interest in this protocol is driven by:
- Social media testimonials
- Celebrity endorsements
- Demand for alternative treatments
- Distrust of conventional therapies
However, many online claims are:
- Unverified
- Selectively reported
- Biased toward positive outcomes
Can It Be Combined with Conventional Cancer Treatment?
At present:
- No established combination protocols exist
- Safety remains uncertain
- Drug interaction risks are present
Use should only be considered under qualified medical supervision.
Ethical and Regulatory Considerations
Major health authorities have not approved these drugs for cancer treatment, including:
- FDA
- EMA
- CDSCO
Off-label use requires:
- Scientific justification
- Informed patient consent
- Close medical monitoring

Drug Repurposing in Cancer: A Broader Perspective
Drug repurposing is a legitimate and growing field in oncology. Benefits include:
- Reduced development costs
- Established safety profiles (for approved uses)
- Faster clinical trial pathways
However, every repurposed drug must undergo rigorous testing before being accepted for cancer treatment.
How Patients Should Evaluate New Cancer Treatments
Before considering any treatment, patients should ask:
- Is it supported by clinical trials?
- Is it approved by medical authorities?
- What are the known risks?
- Has an oncologist recommended it?
Integrative vs Alternative Treatment
- Integrative care combines evidence-based medicine with supportive therapies
- Alternative care replaces conventional treatment
The antiparasitic protocol currently falls under experimental or alternative treatment, not integrative oncology.
Future Research Directions
Researchers continue to explore:
- Drug repurposing strategies
- Combination therapies
- Targeted drug delivery systems
Conclusion
Fenbendazole, ivermectin, and mebendazole show potential anticancer activity in preclinical studies. However, the antiparasitic cancer protocol remains unproven in humans and carries significant safety risks.
Patients should prioritize evidence-based treatments and consult qualified healthcare professionals before considering experimental approaches.
FAQ's
Fenbendazole and Ivermectin have shown anticancer activity in laboratory and animal studies, such as disrupting cancer cell division and triggering apoptosis. They are currently considered experimental in oncology research.
Yes, but they are limited:
- Mebendazole has been studied in early-stage trials (e.g., brain tumors like glioblastoma)
- Ivermectin has small-scale or preliminary studies
- There are no high-quality, large-scale trials proving effectiveness of combination protocols
More research is needed before any medical recommendations can be made.
There is currently:
- No established protocol
- Limited safety data
- Potential for harmful drug interactions
Any such use should only be considered under strict medical supervision.

.svg)



