Mebendazole for Cancer Treatment: Human Protocol, Dosage Cycles, and Detox Binders
Drug repurposing has gained increasing attention in cancer research as scientists investigate whether medications originally developed for other diseases may have anticancer potential. One such drug is mebendazole, a well-known antiparasitic medicine commonly used to treat intestinal worm infections.
Recent laboratory studies, case reports, and early-stage clinical investigations have explored the possibility that mebendazole may exhibit anticancer activity. Some integrative oncology practitioners have also discussed experimental protocols that combine mebendazole with dietary strategies, supportive supplements, and detoxification approaches.
However, it is important to emphasize that mebendazole is not currently approved by regulatory authorities as a standard cancer treatment. Most of the available evidence remains preclinical or early-stage clinical research, and studies are ongoing.
This article reviews the current scientific evidence, proposed mechanisms of action, experimental dosing strategies mentioned in research settings, and the role of detox binders sometimes discussed in integrative oncology approaches.
What is Mebendazole?
Mebendazole is an anthelmintic medication widely used to treat infections caused by parasitic worms such as:
Roundworms
Whipworms
Pinworms
The drug works by disrupting the microtubule structure of parasites, preventing them from absorbing glucose and ultimately leading to their death.
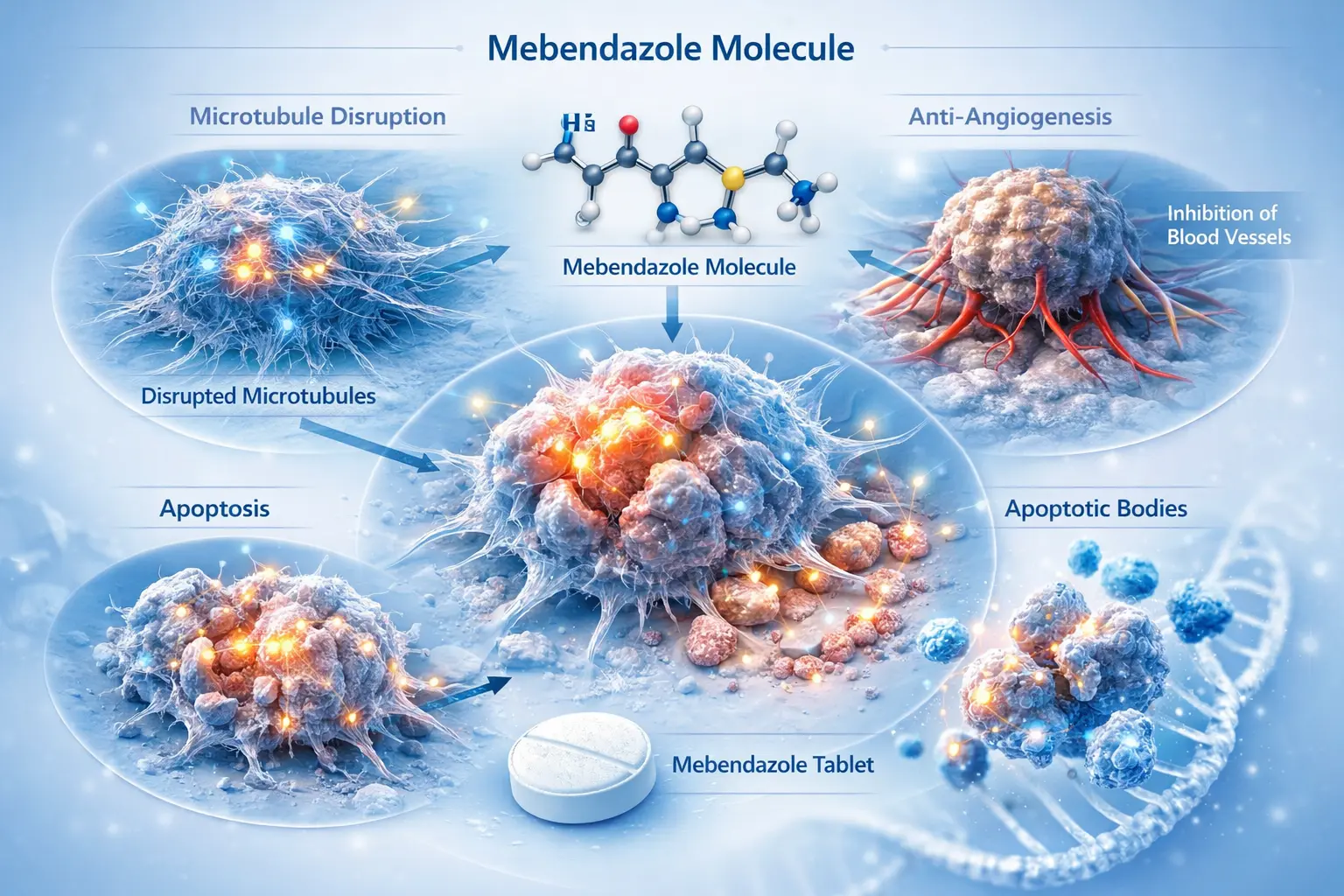
Interestingly, this same mechanism has raised interest among researchers because microtubule disruption may also interfere with the growth of cancer cells.
Potential Anticancer Effects Suggested in Research
Laboratory research suggests that mebendazole may:
Disrupt tumor cell microtubules
Induce programmed cancer cell death (apoptosis)
Inhibit angiogenesis (blood supply to tumors)
Interfere with cancer signaling pathways
Several experimental studies have demonstrated that mebendazole can cause cell-cycle arrest and apoptosis in certain cancer cell lines, suggesting potential anticancer properties.
Because the drug has been used safely as an antiparasitic medication for decades, researchers are exploring whether it may be repurposed as a supportive or adjunct therapy in oncology.
Scientific Evidence from Preclinical Cancer Research
Numerous laboratory and animal studies have investigated the potential anticancer activity of mebendazole.
Findings from Laboratory Studies
Research indicates that mebendazole may:
- Trigger apoptosis in cancer cells
- Prevent cell division by disrupting tubulin formation
- Induce oxidative stress within tumor cells
A study examining breast and colon cancer cell lines found that mebendazole inhibited microtubule formation, leading to cell-cycle arrest and selective killing of cancer cells while sparing normal cells.
Types of Cancer Studied
Preclinical studies have explored the drug's activity in several cancers, including:
- Glioblastoma
- Melanoma
- Lung cancer
- Leukemia
- Colon cancer
Animal model research has also suggested potential benefits. For example, one study found that mebendazole combined with radiation therapy improved survival in brain tumor models.
Human Clinical Research
Human clinical research on mebendazole in oncology remains limited.
A Phase 2a clinical study evaluated individualized dosing of mebendazole in patients with advanced gastrointestinal cancers.
Key Findings from the Trial
Researchers reported that:
- The drug was generally well tolerated
- Daily doses up to 4 grams were administered
- Some participants experienced disease progression during the study
These findings suggest that while mebendazole appears relatively safe at higher doses, its independent anticancer effectiveness in humans is still uncertain.
As a result, researchers are investigating whether mebendazole might work better in combination with other cancer treatments.
Proposed Anticancer Mechanisms of Mebendazole
Scientists believe that mebendazole may influence cancer growth through multiple biological pathways.
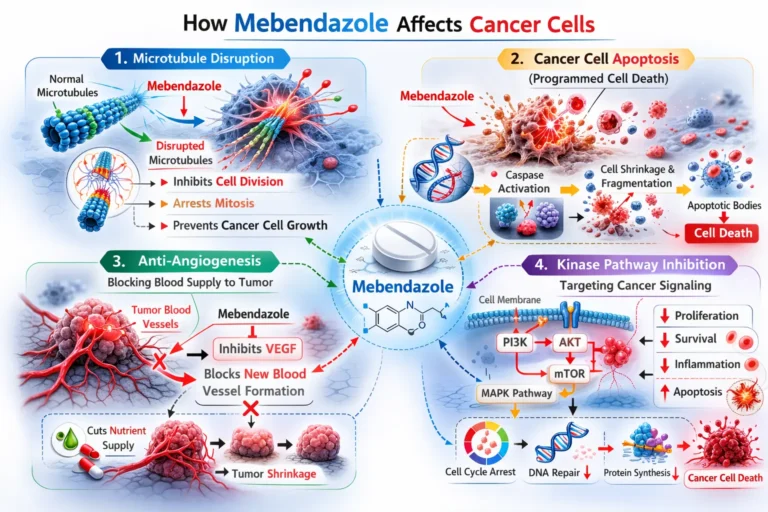
Microtubule Disruption
Mebendazole interferes with tubulin polymerization, damaging the structural components necessary for cell division. Because cancer cells divide rapidly, they are particularly vulnerable to drugs that disrupt microtubules.
Apoptosis Induction
Studies suggest that the drug activates caspase pathways, which trigger programmed cell death in tumor cells.
Anti-Angiogenesis Effects
Tumors require new blood vessels to grow. Research suggests that mebendazole may reduce tumor vascularization, limiting the blood supply required for tumor expansion.
Kinase Inhibition
Some research indicates that mebendazole may inhibit several signaling proteins involved in cancer growth pathways, including:
- BRAF
- ABL1
- PDGFRA
These proteins are often involved in tumor development and proliferation pathways.
Experimental Human Protocols in Integrative Oncology
Some integrative oncology practitioners have discussed experimental mebendazole protocols for human use. These protocols vary significantly and should never be attempted without medical supervision.
Common Components Discussed in Integrative Protocols
Experimental protocols sometimes include:
- Cycles of mebendazole therapy
- Nutritional strategies
- Supportive supplements
- Detoxification approaches
It is important to understand that these approaches are not standardized medical treatments and lack extensive clinical validation.
Experimental Mebendazole Dosage Cycles
In clinical research settings, dose-escalation strategies have been used to study mebendazole.
For example, one clinical trial adjusted dosages up to 4 grams per day in order to reach specific blood concentration levels.
In some integrative oncology settings, lower doses may be used in cycles.
Example of an Experimental Cycle
Commonly discussed patterns may include:
- Daily dosing
- 100–200 mg per dose
- Taken two or three times per day
- Consumed with high-fat meals to improve absorption
Again, these protocols are not standardized and should only be evaluated under medical supervision.
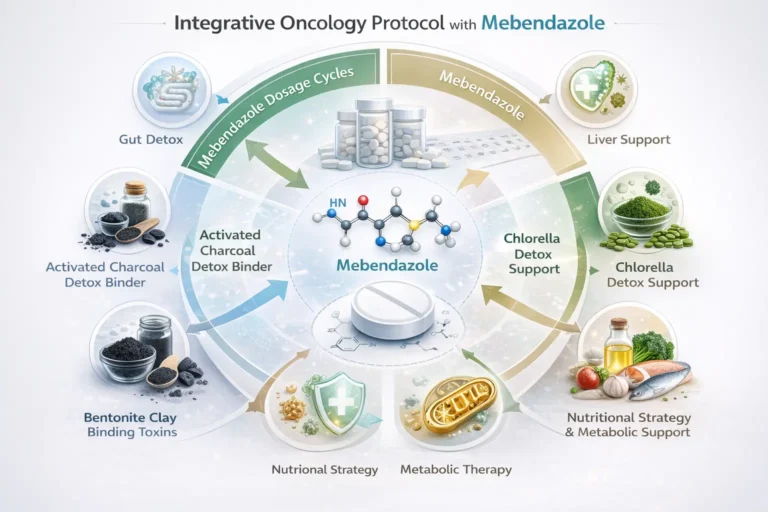
Detox Binders in Integrative Cancer Protocols
Some integrative treatment protocols include detox binders, substances designed to bind toxins or metabolic byproducts within the digestive tract. These substances are sometimes discussed in alternative or integrative oncology approaches aimed at supporting the body during certain experimental treatment strategies.
Why Detox Binders May Be Used
Supporters of this approach suggest several possible reasons for including detox binders in experimental protocols:
Tumor cell death may release inflammatory compounds into the body
Antiparasitic drugs may increase toxin elimination
Gut detoxification may improve treatment tolerance in some individuals
However, it is important to note that the use of detox binders in cancer therapy remains controversial within conventional oncology, and scientific evidence supporting these practices is still limited.
Common Detox Binders Discussed in Integrative Protocols
Activated Charcoal
Activated charcoal is widely known for its ability to bind toxins and chemical substances in the digestive tract. It has historically been used in emergency medicine for certain types of poisoning due to its strong adsorption properties.
Suggested benefits discussed in integrative protocols include:
Reduction in toxin absorption within the gut
Support for digestive detoxification processes
However, clinical evidence supporting its routine use in cancer-related detox protocols remains limited.
Bentonite Clay
Bentonite clay is a naturally occurring mineral clay that is believed to bind toxins, chemicals, and heavy metals in the digestive tract.
In integrative health discussions, it is sometimes used as part of detox programs. However, scientific data supporting its use in cancer detox protocols is still minimal, and further research is needed.
Chlorella
Chlorella is a nutrient-dense green algae supplement often used in nutritional and detoxification programs.
Some proposed benefits include:
Binding of certain heavy metals
Potential immune system support
Despite these claims, clinical research validating chlorella for cancer detoxification remains limited, and more studies are required.
Possible Side Effects and Safety Concerns
When used for treating parasitic infections, mebendazole is generally considered safe and well tolerated. However, higher doses or prolonged use may lead to certain side effects.
Potential Side Effects
Possible adverse effects may include:
Elevated liver enzyme levels
Stomach discomfort
Abdominal pain
Rare cases of neutropenia
Allergic reactions
In one clinical study, some participants experienced increased liver enzyme levels, which required dosage adjustments. Because of these potential risks, medical supervision and routine monitoring are recommended.
Who Should Avoid Mebendazole?
Off-label use of mebendazole should only be considered under medical supervision. Certain individuals may need to avoid the medication unless advised by a qualified healthcare professional.
Individuals Who Should Exercise Caution
This may include:
Pregnant women
Individuals with severe liver disease
Patients taking medications that may interact with mebendazole
Immunocompromised individuals not under oncologist supervision
Proper medical evaluation is essential before considering any off-label therapeutic use.
Current Role of Mebendazole in Cancer Research
At present, mebendazole remains an experimental compound in oncology research. Scientists are continuing to explore its potential applications through laboratory studies and clinical trials.
Current research is evaluating whether mebendazole may function as:
A radiosensitizer
A component of metabolic cancer therapies
A repurposed oncology medication
A supportive treatment alongside chemotherapy
However, more randomized clinical trials and large-scale human studies are required before mebendazole could be considered a standard treatment option in cancer care.
FAQ'S
It may increase the efficacy of radiation or chemotherapy, according to some study, although this is still experimental and should only take place under medical supervision.
In integrative medicine, detox binders are occasionally used to lessen the absorption of toxins during treatment. Nevertheless, there is little evidence to support their use in cancer treatment.
For parasites, short-term usage is usually safe. Medical monitoring of blood counts and liver function may be necessary for long-term or high-dose use.
No, mebendazole is not licensed for cancer, but rather for parasitic illnesses. To assess possible anticancer effects, research is still being conducted.
Individualized dosages up to 4 grams daily have been utilized in clinical trials; however, dosing varies based on patient monitoring and study design.

.svg)



