Research and Protocol Differences
There has been a significant increase in interest in drug repurposing in cancer research. Drug repurposing is the process of examining the currently available drugs that have been approved for a particular indication in order to regulate whether they may show therapeutic potential for other illnesses. Mebendazole vs Fenbendazole for Cancer, two members of the benzimidazole class that are typically employed as antiparasitic treatments, are among the medications that have gathered interest in laboratory cancer research.
Mebendazole vs. Fenbendazole for Cancer
Mebendazole and fenbendazole are members of the benzimidazole class of chemicals. These medicines were first used to treat parasitic worm infections by preventing helminths from forming micro tubules. Cell division, intracellular transport, and shape preservation depends on microtubules, which are the structural elements of cells.
For cancer cells to multiply, microtubule activity must be intact. Because of this biological overlap, scientists are exploring whether benzimidazoles may exhibit anticancer action through comparable pathways. It is crucial to stress that clinical efficacy is not the same as mechanistic plausibility. Due to pharmacokinetic restrictions, toxicity, or a lack of repeatable benefit, several substances show anticancer potential in lab conditions but fail in human trials.
Mebendazole: Research Background and Regulatory Status
Mebendazole can cause mitotic arrest and death in cancer cells by interfering with tubulin polymerization, according to preclinical research. Mebendazole is discussed as a potential cancer medication repurposing in a thorough study that was published in the journal Cancers.
Many nations have approved mebendazole for use as an antiparasitic drug in humans. Researchers have investigated its potential for therapeutic repurposing in oncology because it has a proven safety record at recommended levels for parasitic illness.
Benzimidazole-treated breast and colon cancer cell lines showed selective apoptotic cell death. While these results are intriguing from a scientific standpoint, the majority of the data are preclinical, meaning they were produced in animal models or cell cultures rather than extensive randomized human clinical trials.
Fenbendazole: Emerging Laboratory Data
Although Mebendazole vs Fenbendazole for Cancer are chemically related, fenbendazole is mostly authorized for use in veterinary medicine. The U.S. Food and Drug Administration has not licensed it for use in humans. But it is frequently used in animals to treat parasite infections. Because veterinary formulations are not produced in accordance with the same regulatory criteria as human medications, this divergence has medicinal significance.
Fenbendazole functions as a moderate microtubule destabilizing agent and promotes cancer cell death by altering several cellular pathways. The possible effects of fenbendazole on tumor models, such as cervical cancer cell systems, have been studied in lab studies that are indexed in PubMed. Fenbendazole has not yet been shown to improve clinical outcomes or survival in cancer patients in any sizable, high-quality randomized controlled studies.
Following research in the lab that indicated fenbendazole might disrupt the microtubules and glucose metabolism of cancer cells, interest in the drug in oncology increased. Fenbendazole functions as a moderate microtubule destabilizing agent and modifies several biological pathways linked to cancer cell survival.
Microtubule disruption is the mechanism of action
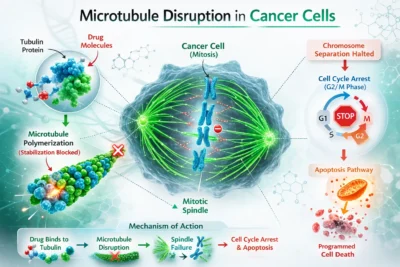
The fundamental mechanism of Mebendazole vs Fenbendazole for Cancer is the interruption of microtubule polymerization. Tubulin proteins make up microtubules, which are necessary for mitosis. Microtubules create the mitotic spindle, which divides chromosomes into daughter cells during cell division.
According to lab tests, mebendazole attaches itself to tubulin and stops it from properly polymerizing. In vitro, fenbendazole seems to exhibit comparable action. However, given variations in absorption and metabolism, in vitro efficacy does not ensure adequate amounts in human tumors when taken orally.
Cells may stop in the G2/M phase of the cell cycle if microtubule assembly is disturbed. Apoptosis, a type of programmed cell death, can be brought on by prolonged arrest. Although benzimidazoles differ in potency, pharmacodynamics, and clinical data, this process is theoretically comparable to that of certain well-known chemotherapy drugs, such as vinca alkaloids and taxanes.
Impacts on Cellular Energy and Glucose Metabolism
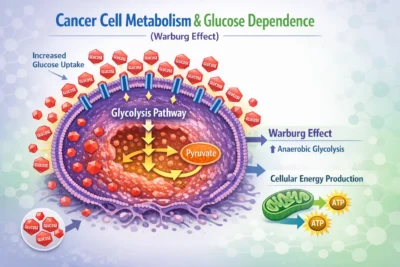
According to certain research, fenbendazole may prevent cancer cells from absorbing glucose in addition to disrupting microtubules. Increased glucose consumption is often necessary for cancer cells to proliferate quickly; this phenomenon is known as the Warburg effect. Theoretically, tumor growth could be hindered by inhibition of glycolysis or glucose transporters.
The impact of fenbendazole on many pathways, including metabolic activities, is described in the Scientific Reports article already mentioned. Despite being mechanistically intriguing, these results are still limited to lab settings. There is not enough clinical data to draw the conclusion that these metabolic alterations have a significant impact on cancer patients in humans. Mebendazole’s possible effects on angiogenesis the process by which tumors create new blood vessels to support growth have also been investigated. Again, strong human validation is missing, while some preclinical models indicate anti-angiogenic qualities.
The Risks that are associated with self-administration of veterinary products include inconsistent dosage, unclear quality, contamination, and a lack of medical supervision. High doses of benzimidazoles are linked to liver damage in some situations, which emphasizes the necessity of medical supervision.
Mebendazole vs Fenbendazole for Cancer will often differ from a regulatory perspective. Mebendazole has been approved for parasitic infections after being assessed in humans. Fenbendazole’s safety profile in people has not been thoroughly examined in controlled clinical trials, nor has it been authorized for use in humans.
Present Clinical Evidence
Randomized controlled trials are the gold standard for validating cancer treatments. There are currently few early phase or exploratory studies using mebendazole in oncology, but there is not any comprehensive, conclusive data showing better disease control or survival in the main cancer types.
Acknowledgment of ambiguity, a clear regulatory framework, transparent sourcing, and unambiguous disclaimers are all necessary for responsible reporting. Major oncology recommendations do not yet accept mebendazole or fenbendazole as conventional cancer therapies. There is considerably less clinical evidence for fenbendazole. Instead of using peer-reviewed randomized trials, a lot of the discussion in public forums is based on anecdotal reports. Anecdotes cannot prove safety, efficacy, or causation.
Conclusion
Important scientific and regulatory differences are highlighted in Mebendazole vs Fenbendazole for Cancer: Mechanism, Research, and Protocol Differences. Both medications have anticancer effects in cell and animal models and exhibit microtubule-disrupting characteristics in lab settings. While fenbendazole is still a veterinary drug without official human approval, mebendazole has the benefit of documented human safety evidence for parasite usage.
There is not enough high-quality clinical evidence to support either medication as a conventional cancer treatment, despite encouraging preclinical results. Patients should communicate any interest in research with qualified healthcare providers and rely on evidence-based oncology therapy.
Frequently Asked Questions
In lab models, both the medications are thought to cause cell cycle arrest and death by interfering with the production of microtubules in cells.
Mebendazole has been the subject of a few early studies, however there is no solid evidence from large-scale randomized trials showing a therapeutic benefit.
Self-medication is not advised for patients. Only under the guidance of a professional oncologist or in an authorized clinical trial setting should any off-label or investigational therapy be taken into consideration.
Yes, it is approved for the treatment of several parasitic illnesses, but not for the treatment of cancer.
The use of fenbendazole in humans is prohibited. Controlled clinical trials have not sufficiently confirmed its safety, dosage, and long-term effects in humans.

.svg)



