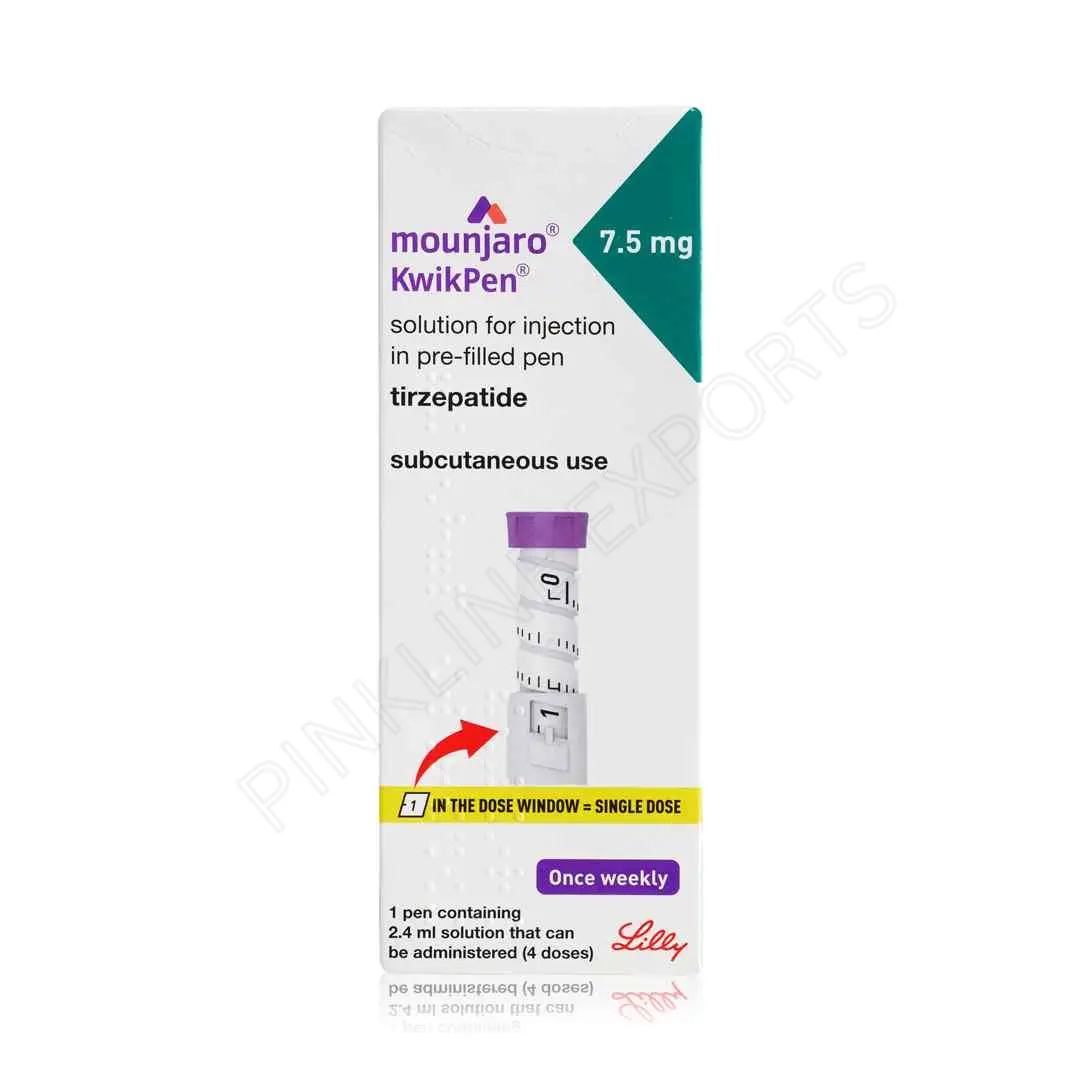
Mounjaro 7.5 Mg – Kwikpen
$320.00
Mounjaro 7.5 Mg – Kwikpen is a once-weekly injectable medication used for the management of type 2 diabetes in adults. It contains tirzepatide and is administered through a prefilled Kwikpen designed for accurate dosing and ease of use. This dosage strength is typically prescribed for patients who have already tolerated lower doses and require enhanced glycemic control.
Description
Mounjaro 7.5 Mg – Kwikpen is an advanced injectable therapy developed to support effective blood glucose management in adults with type 2 diabetes mellitus. As diabetes continues to be a growing global health concern, modern treatment options such as Mounjaro offer a multi-pathway approach to glycemic regulation, addressing both insulin secretion and appetite control.
The active pharmaceutical ingredient, tirzepatide, is a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. This dual mechanism differentiates Mounjaro from traditional incretin-based therapies and contributes to its strong clinical efficacy.
The 7.5 mg strength is part of the step-up dosing regimen and is generally prescribed after successful initiation with lower doses. Mounjaro 7.5 Mg – Kwikpen is not intended for first-time users but rather for patients who require improved glycemic outcomes after dose escalation under medical supervision.
Mechanism of Action
Mounjaro works by mimicking natural incretin hormones released by the gut in response to food intake. Tirzepatide activates both GIP and GLP-1 receptors, resulting in:
-
Increased insulin secretion when blood glucose levels are elevated
-
Reduced glucagon release, lowering hepatic glucose production
-
Delayed gastric emptying, helping control post-meal glucose spikes
-
Increased satiety, which may support weight management
The combined hormonal activity enhances metabolic regulation and allows Mounjaro 7.5 Mg – Kwikpen to provide sustained glucose control throughout the week.
Indications and Therapeutic Use
Mounjaro 7.5 Mg – Kwikpen is indicated for:
-
Adults diagnosed with type 2 diabetes mellitus
-
Patients requiring intensified glycemic control after initial therapy
-
Use as an adjunct to diet modification and increased physical activity
It is not indicated for patients with type 1 diabetes or for the treatment of diabetic ketoacidosis. Prescription and dose progression should always be determined by a qualified healthcare professional.
Dosage and Administration
The recommended dose of Mounjaro 7.5 Mg – Kwikpen is one subcutaneous injection once weekly.
Administration guidelines include:
-
Inject on the same day each week
-
Injection sites include the abdomen, thigh, or upper arm
-
Rotate injection sites to minimize local reactions
-
Can be administered with or without food
The Kwikpen delivery system ensures precise dosing without the need for manual measurement, improving patient confidence and adherence.
Benefits of Mounjaro 7.5 Mg – Kwikpen
Patients using Mounjaro 7.5 Mg – Kwikpen may experience several clinical and practical benefits, including:
-
Enhanced blood glucose control compared to lower doses
-
Once-weekly dosing convenience
-
Dual-action incretin therapy
-
Potential support for weight reduction when combined with lifestyle changes
-
Reduced treatment burden through prefilled pen administration
This dosage strength is particularly beneficial for patients who have not achieved target glycemic goals with earlier dose levels.
Safety Information and Warnings
Before starting Mounjaro 7.5 Mg – Kwikpen, patients should disclose their full medical history to their healthcare provider.
Key safety considerations include:
-
History of pancreatitis
-
Personal or family history of medullary thyroid carcinoma
-
Multiple endocrine neoplasia syndrome type 2
-
Severe gastrointestinal disorders
Common side effects may include nausea, vomiting, diarrhea, constipation, and decreased appetite. These effects are generally dose-dependent and may lessen with continued use.
Serious adverse reactions are uncommon but require immediate medical attention if they occur.
Drug Interactions
Due to delayed gastric emptying, Mounjaro may affect the absorption of orally administered medications. Caution is advised when taken with drugs that require rapid gastrointestinal absorption.
When combined with insulin or insulin secretagogues, dose adjustments may be required to reduce the risk of hypoglycemia.
Always consult a healthcare provider before combining Mounjaro 7.5 Mg – Kwikpen with other antidiabetic therapies.
Storage and Handling Instructions
To maintain product quality and effectiveness:
-
Store refrigerated at 2°C to 8°C before first use
-
Do not freeze
-
Keep away from direct heat and sunlight
-
Once opened, follow healthcare guidance for room-temperature storage
-
Keep out of reach of children
Improper storage may reduce the effectiveness of Mounjaro 7.5 Mg – Kwikpen.
Frequently Asked Questions (FAQs)
1. What is Mounjaro 7.5 Mg – Kwikpen used for?
It is used to improve blood sugar control in adults with type 2 diabetes who require dose escalation.
2. Is this dose suitable for beginners?
No, this strength is typically prescribed after successful use of lower doses.
3. How often is the injection taken?
Once weekly on the same day each week.
4. Can this dose help with weight management?
Some patients may experience weight reduction, although it is not approved solely for weight loss.
5. Is Mounjaro safe for long-term use?
Long-term use should be monitored by a healthcare professional.
6. Does it cause low blood sugar?
The risk is low when used alone but may increase with other antidiabetic medications.
7. Can injection days be changed?
Yes, provided at least 72 hours have passed since the previous dose.
8. What happens if a dose is missed?
If missed within 4 days, take it as soon as possible. Otherwise, skip and resume the schedule.
9. Does the pen need refrigeration?
Yes, refrigeration is required before initial use.
10. Who should avoid Mounjaro?
Patients with type 1 diabetes or specific thyroid conditions should not use it.
Conclusion
Mounjaro 7.5 Mg – Kwikpen is a clinically advanced option for adults with type 2 diabetes who require stronger glycemic control following initial therapy. Its dual GIP and GLP-1 receptor mechanism, combined with a convenient once-weekly Kwikpen format, supports improved metabolic outcomes and long-term treatment adherence. When used under proper medical guidance, it plays a significant role in modern diabetes management strategies.

.svg)







Reviews
There are no reviews yet.